Parenting(Age 5 to 8) | Academic | General | Parenting(Age 9 to 12) | Parenting(Age 13 to 16) | Sep 15, 2021
What Are The Seven Basic Units Of Measurement?
Basic, fundamental, or standard, whatever you may call it when speaking for measurement units, you will land at the same seven units. Whether Physics, Chemistry, or Mathematics, the seven fundamental units of measurement play a crucial role in the subjects. As the name suggests, these are the units for measuring different kinds of physical and non-physical quantities. They get described as a definite magnitude of a quantity. These units are defined and adopted by the convention and are used as a standard for the measurement of the same kind of quantities.
Get Free Consultation Now!
What is a standard unit of measurement?
A standard measurement unit is a quantifiable language used for describing the magnitude of a quantity. It builds an association of the object with the measurement and helps us to understand this association. Measurement is a significant part of our lives. Therefore, kids must understand the measuring units.
Why are Standard units needed?
There are several systems and units for measuring different quantities. Now, imagine what would happen if one country utilizes one unit for measuring the size or amount of an object while the other uses a different one for the same thing? It would create confusion and make it difficult for the two countries to communicate for import and export or any and every other purpose. Hence, a standard system of units was developed for the uniformity of measurement all around the world. All the other units can be converted into SI units for mutual understanding.
What is the International System of Units?
In ancient times, people did not have any devices or units for measuring the number or quantity of objects. To tackle this, people came up with various methods for measuring. They started using the tools available at hand. For example, palm and foot measurement methods were used for the length. But, these methods were not accurate. Hence, a system of units called the International System of Units was developed. The system is now being used by almost all developed and developing countries. The SI units are being used across all fields like science and technology and in government operations.
Who decides Standard units?
The right for authorizing a standard unit is reserved with a body named GCPM- General Conference on Weight and Measures. The authority ensures that the units will be accepted internationally and not abrupt communication between two countries, affecting their economy. Changes in the standard units are announced by the organization through meetings. They also address the changes through publications.
How are Derived units different from Fundamental units?
The derived units are formed by the product of the powers of the fundamental units. These quantities derived from the standard units. The basic units can be independently defined. But, the derived units are not independently defined, as they depend on the definition of the fundamental units.
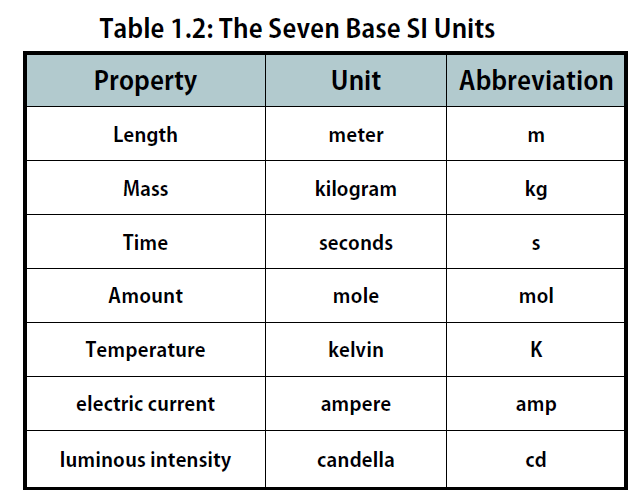
Seven Basic units of measurement
Each unit of the standard units is dimensionally independent of the others and are as follows-
Kilogram (Kg)
The SI unit of mass is defined by the mass of a platinum-iridium cylinder prototype at the International Bureau of Weights and Measures in Paris, France.Metre (m)
The SI unit for length is meters. The distance traveled by the light in 1/299,792,458 seconds is equal to one meter.Second (s)
For time, the standard unit is second. It is described as the time taken to complete 9,192,631,770 oscillations by radiations corresponding to the transition between the two hyper fine levels of the Caesium-133 atom.Ampere (A)
The basic unit for electric current is Ampere. One Ampere is the constant current maintained in two infinitely long straight parallel conductors. The circular cross-section is negligible here, and the conductors get placed one meter apart, producing a force between the conductors equal to 2 x 10-7 newtons per meter of length.Kelvin (K)
It is the SI unit of temperature, defined as the fraction 1/273.16 of the thermodynamic temperature of the triple point of water. The Kelvin scale is taken as an absolute scale. Hence it does not have a degree.Mole (mol)
For measuring the amount of a substance, the basic unit is a mole. One mole is the amount of substance equal to the atoms in 0.012 kilograms of a Carbon-12 atom.Candela (cd)
The SI unit for luminous intensity is the candela (cd). It is equal to the luminous intensity of an origin emitting monochromatic radiation of 540 x 1012 hertz with a radiant level in the same region of 1/683 watt per steradian.
Wrapping Up!
The seven basic units of measurement hold a valuable place in the world. From the problems in school to the astronomy world, these units are used uniformly. All the mentioned quantities have other units too, which can get converted into SI units. Recently some changes were made in these seven units by CGPM in 2019
- The fundamental unit of mass- kilogram will be based on the Planck constant, and for this, a Kibble Balance will be utilized.
- Electron's charge will be used to define Ampere.
- For defining a mole, Avogadro Constant will be used.
- Kelvin must be based on the Boltzmann Constant.















Post a Comment: